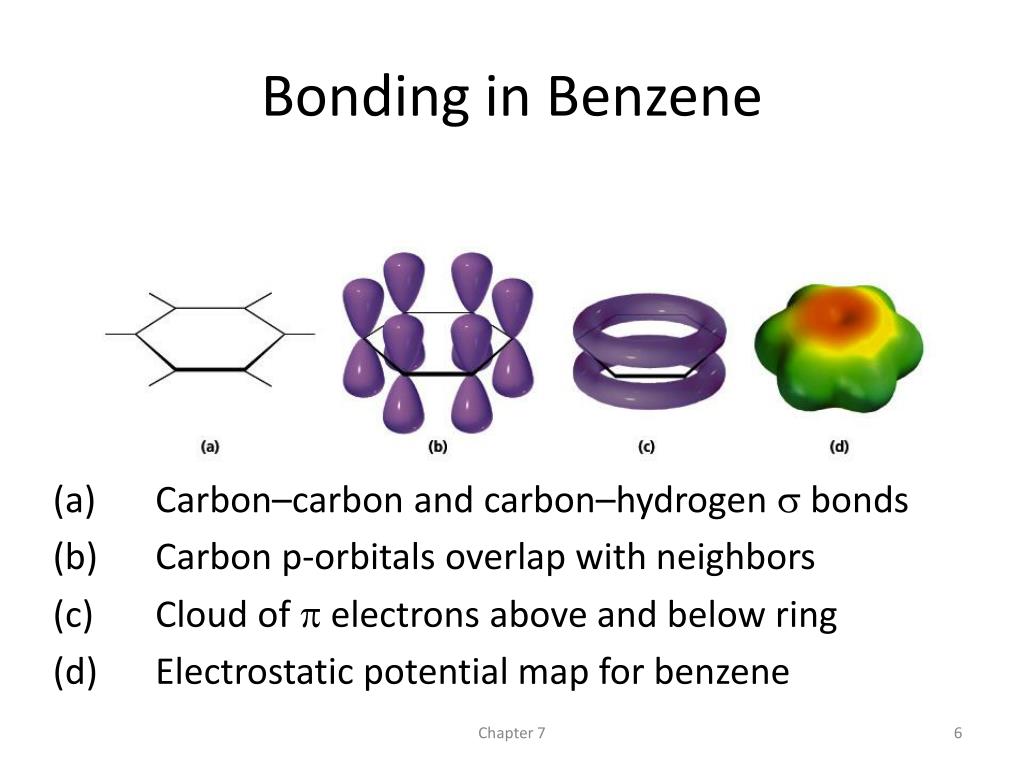
As Benzene is a cyclic hydrocarbon containing 3 double bonds the hydrogenation of Benzene should be around -360 kJmol -1. Cyclohexane is a cyclic hydrocarbon containing 1 double bond and its energy of hydrogenation is well-known to be -120 kJmol -1. The energy of Hydrogenation, which is the addition of Hydrogen, did not fit with the idea of 3 double bonds either. This was not the only problem with Kelulé’s proposed structure of Benzene. In reality both of these structure are identical and so this was not a valid explanation. Kekulé tried to explain this by saying that Benzene was in a dynamic equilibrium where the double bonds changed position constantly and therefore there was not time for the Bromine to react with the double bond before it changed position again. (3) This caused massive confusion within the scientific community as no one could understand why it didn’t react as its structure should. Despite this sound theoretical chemical reasoning Benzene did not react with bromine in the way that other molecules with double bonds do, it rarely reacted and if it did it was a substitution reaction not addition. They proposed that in dark conditions at room temperature Benzene would undergo an electrophilic addition reaction with bromine. The remaining bond on each carbon was joined to a hydrogen and this is where the 6 hydrogen atoms were used.Īs the molecule contained 3 double bonds, chemists expected that it would react in much the same way that alkenes do, only more so due to the increased number of bonds. This meant the molecule had 3 single and 3 double bonds making 3 bonds for each carbon atom. (2) He proposed that the 6 Carbon atoms were arranged in a regular Hexagon joined by alternating single and double bonds. This is where Kelulé’s idea for the cyclic structure of Benzene came from. He had a dream in which a snake was eating its own tail. The first structure that was considered to be a valid possibility was one proposed by German organic chemist Friedrich August Kekulé. Many scientists tried to work out the structure of this molecule to little success. However, Benzene turned out to be a surprisingly unreactive molecule. 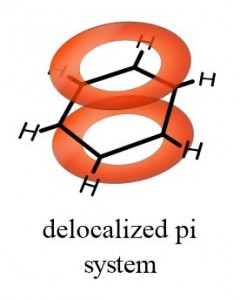
(1) The formula of C 6H 6 implied that the structure would contain several double bonds and would therefore be incredibly reactive. The molecule Benzene was first isolated as a hydrocarbon by Michael Faraday in 1825.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
